Innovative SDT Treatment Approaches
Healthcare Professionals
Your call
Your Call to Action...
We are not yet recruiting but will update the website and contact relevant neuro-oncologists when we have the necessary permissions and processes in place to start. Watch this space.
Trials
Drs. and Nurses
Patients
Years of Experience
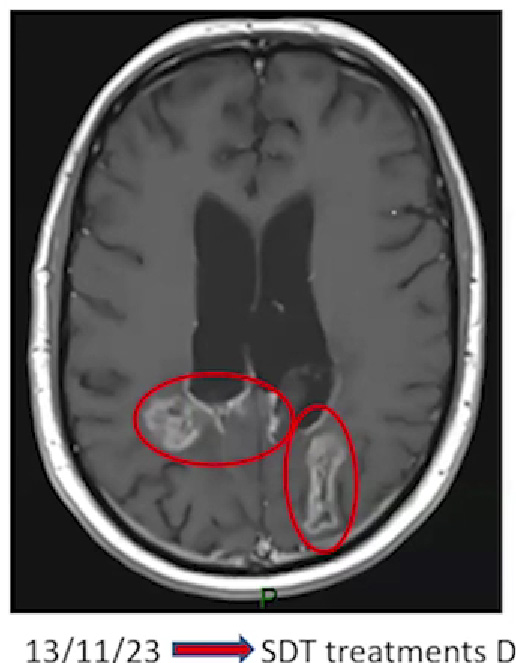
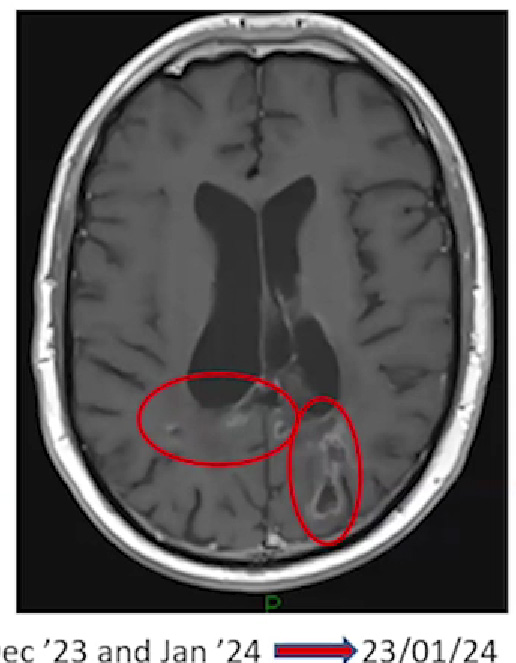
Duncan Campbell's brain scan
Read Duncan's (27 years) full story. Despite being diagnosed with glioblastoma in February 2021, he is living his best life, with a successful career and a fulfilling social life.
BACKGROUND
primary brain tumour
Glioblastoma (GBM) is the most frequent malignant primary brain tumour in adults. Despite standard of care (SOC) treatment with maximal resective safe surgery, radiotherapy and chemotherapy, median overall survival from diagnosis is only 14-15 months (or 6-8 months from tumour recurrence (recGBM) after SOC). There is an unmet need for safe and minimally invasive interventions that could treat GBM rapidly, when recurrence is noted in 3 monthly MRI surveillance scans.
Sonodynamic therapy offers a means to combat the unique heterogeneity of GBM, as well as its diffuse infiltrative nature that makes it so difficult to treat. It involves ingestion of a ‘sonosenitiser’ drug (5-ALA) that leads to accumulation of its active metabolite, PPIX, in cancerous cells, no matter what their molecular subtype is – i.e. it will accumulate in cancer cells that have survived and are resistant to radiotherapy and chemotherapy (with >95% sensitivity – no chemotherapeutic offers this degree of ‘precision medicine‘).
ultrasound energy
Delivering focused ultrasound energy promotes selective cell kill through generation of reactive oxygen species, whilst sparing surrounding normal brain tissue: sonodynamic therapy (SDT) (1). Early safety studies with other ultrasound-delivery devices have yielded incredibly promising results with a doubling of median survival to 15 months for patients with recGBM (4,11) and 16 months for DIPG (a paediatric brainstem tumour with median life expectancy of 9mths).
RATIONALE
No novel agent has added more than 3-4 months life expectancy to people with GBM. The lack of local or systemic toxicity observed after trials of SDT in malignant brain tumours, and the lack of efficacy of current first line therapy for GBM, motivated us to find a fully navigatable FUS device – the potential new treatment device uses over 400 220KHz ultrasound transducers to focus energy to any part of the brain (US Patent 18/146,028) and therefore focally treat recurrent GBM. This potential new treatment device has received U.S. FDA Fast Track Designation for use in human liquid biopsy tests and has been shown to be safe in a successful human efficacy trial (2), with a further 40 patients to be recruited in a new trial of sonobiopsy: https://clinicaltrials.gov/study/NCT05281731. It was designed to open the blood brain barrier but, like the FDA-approved 220Khz commercially available Exablate Type 2.0 FUS device, it can also be used to activate ALA and non-invasively treat brain tumours via SDT (3,5,6).
The sensitivity of SDT to selectively kill cancer cells whilst sparing normal brain tissue may explain the lack of serious adverse events (SAEs) seen thus far in early clinical safety studies. Moreover, there is little treatment burden for the patient, as they are awake and without the need for general anaesthesia during their outpatient treatment. SDT will not lead to postponement of any second-line / salvage adjuvant therapy. As a substantial additional benefit, SDT works as a potential auto-immune vaccine via its abscopal effect. This hypothesis is based on clinical trials using laser light to activate ALA, which showed that not only was the targeted tumour destroyed by ALA activation but also satellite and distant lesions, suggesting that the tumour antigens released by photodynamic therapy (PDT) are recognised by the immune system, in the same way that vaccines also cause an immune response. We believe that SDT works by the same pathological processes as PDT. The abscopal effect is discussed in more detail in a review by Collins et al. (9).
At the time of writing, 40+ adults and 15 children have already been treated with SDT for recGBM (e.g. 3,4,7,11i) and DIPG (5) respectively, with no adverse events found due to the SDT therapy.
Secondary end points of the trial are to assess radiological evidence of cell kill (with MRI scans performed the day after SDT treatment and monthly for three further months), progression free survival and overall survival.
Our study aims to prospectively investigate whether SDT can be integrated into the existing care pathway of glioblastoma patients undergoing routine MRI surveillance after completion of the SOC Stupp protocol (8).
Data obtained from our patient participation and inclusion (PPI) survey suggests that people with GBM would be willing to undertake an outpatient procedure such as SDT in addition to their 3 monthly MRI scans, especially as early results show a doubling in survival time for recGBM (4). Feedback was positive for SDT being a non-invasive outpatient procedure, using existing MRI scans and the new potential treatment device offers the benefit of being cheap and portable enough to allow a large network of hospitals to provide SDT close to home, reducing travel time that is a considerable burden for patients and their family / carers – particularly if they have a neurological deficit. Moreover, SDT has been proved safe in children with DIPG (5), unlike many experimental chemotherapeutics which are often not even tested in children. Thus, SDT is being investigated for the whole population – including those deemed too medically unfit or too frail for chemotherapy, as the treatment is so gentle.
RISKS
As no SAEs are yet to be reported with SDT, there is only a theoretical risk of seizures, intra-tumoural swelling and haemorrhage (with subsequent neurological deterioration in the first few hours after treatment). There are theoretical risks with the use of ALA too (allergic reaction, abnormal liver function and sunburn to exposed skin), However, all patients will have had their initial tumour debulking surgery with the use of 5-ALA as a fluorescent-guided tool (which means that we know the 5-ALA will accumulate in their recGBM and that they have not suffered side effects from it previously). Patients will be monitored closely for side effects / SAEs and advised to wear SPF50 and avoid direct sunlight to exposed skin for 24hrs after taking ALA. However, even though these SAEs have never yet been seen, we have informed the Ethics board that the study objectively poses more than minimal but still justifiable and controlled risks.
REDO SURGERY
If a patient requires redo surgery as SOC, they will not be excluded from the trial and SDT will be offered 1-2 days prior to their scheduled operation. This has the added benefit of allowing us to collect spare biopsied tissue to look for histological markers of cell death, as an additional end point of the trial.
Sex assigned at birth and gender will be recorded and considered in the analysis, as biological differences may influence glioblastoma progression and treatment response. Inclusive language will be used in all participant-facing materials, and no exclusion will be made based on sex or gender.
STAGE OF TRIAL
We have completed a trial protocol (see flow chart below) and secured funding for a new potential treatment device purchase and its QA tests to allow use in patients with GBM. Ethical approval for a single site feasibility trial in Bern will be submitted in Q2 2026, with the aim of starting the trial by the end of 2026.
BE PART OF THE BREAKTHROUGH
Study Flow Chart for Feasibility Study of Treatment Device for recGBM
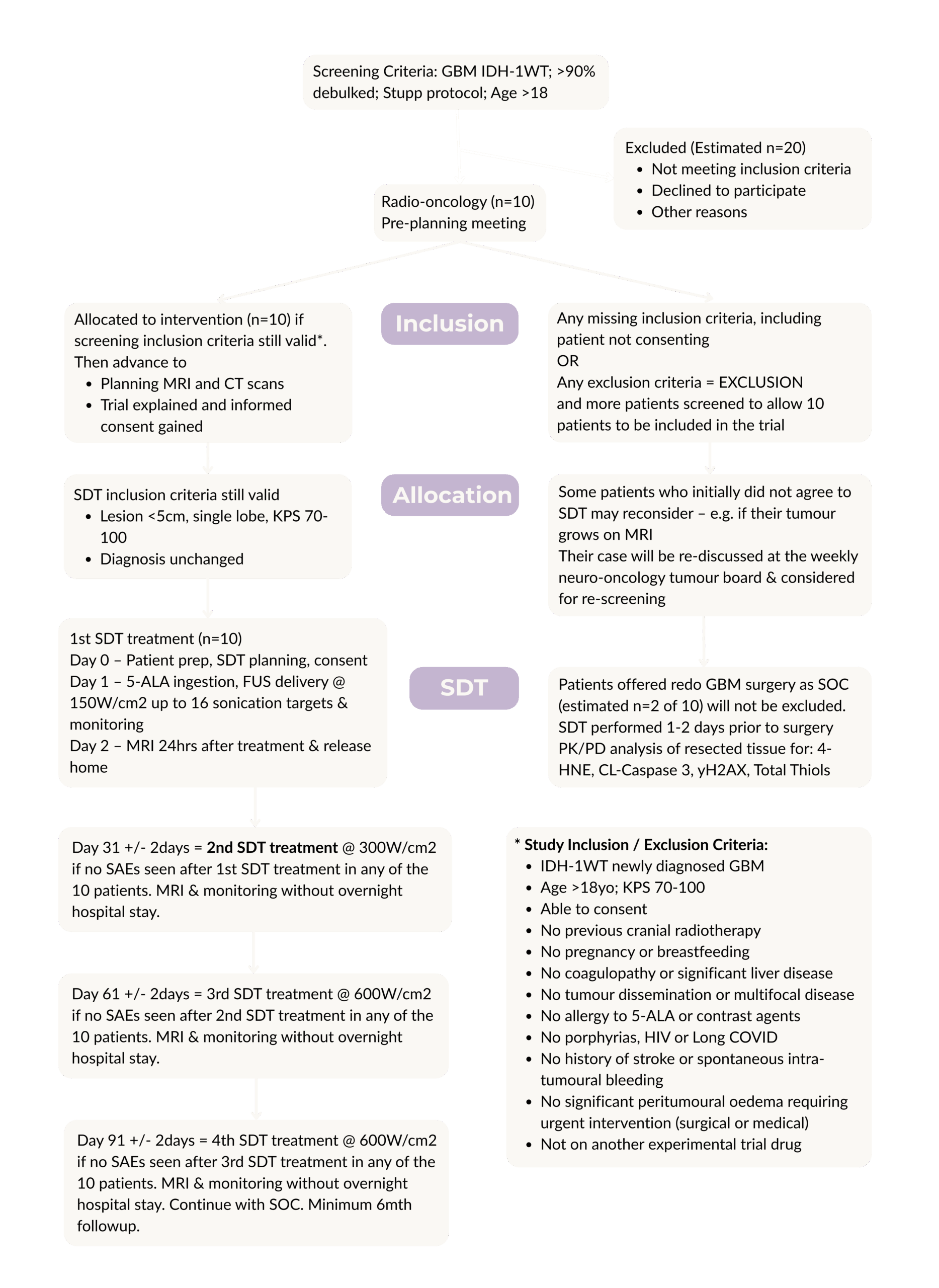
STUDY INCLUSION / EXCLUSION CRITERIA
- IDH-1WT rec GBM on Stupp protocol
- Age >22yo; KPS 70-100
- Able to consent
- Cumulative RT dose <64Gy
- No pregnancy or breastfeeding
- No coagulopathy or significant liver disease
- No dissemination or multifocal disease
- No allergy to 5-ALA or contrast agents
- No porphyrias, HIV or Long COVID
- No history of stroke or spontaneous intra-tumoural bleeding
- No significant peritumoural oedema requiring urgent intervention (surgical or medical)
- Not on another experimental trial drug
Through evaluating feasibility and safety, this study will produce generalisable evidence to inform the design of future randomised or controlled trials.
