References
Information for Researchers
References: Existing Evidence for Safety and Efficacy of Sonodynamic Therapy for Malignant Brain Tumours
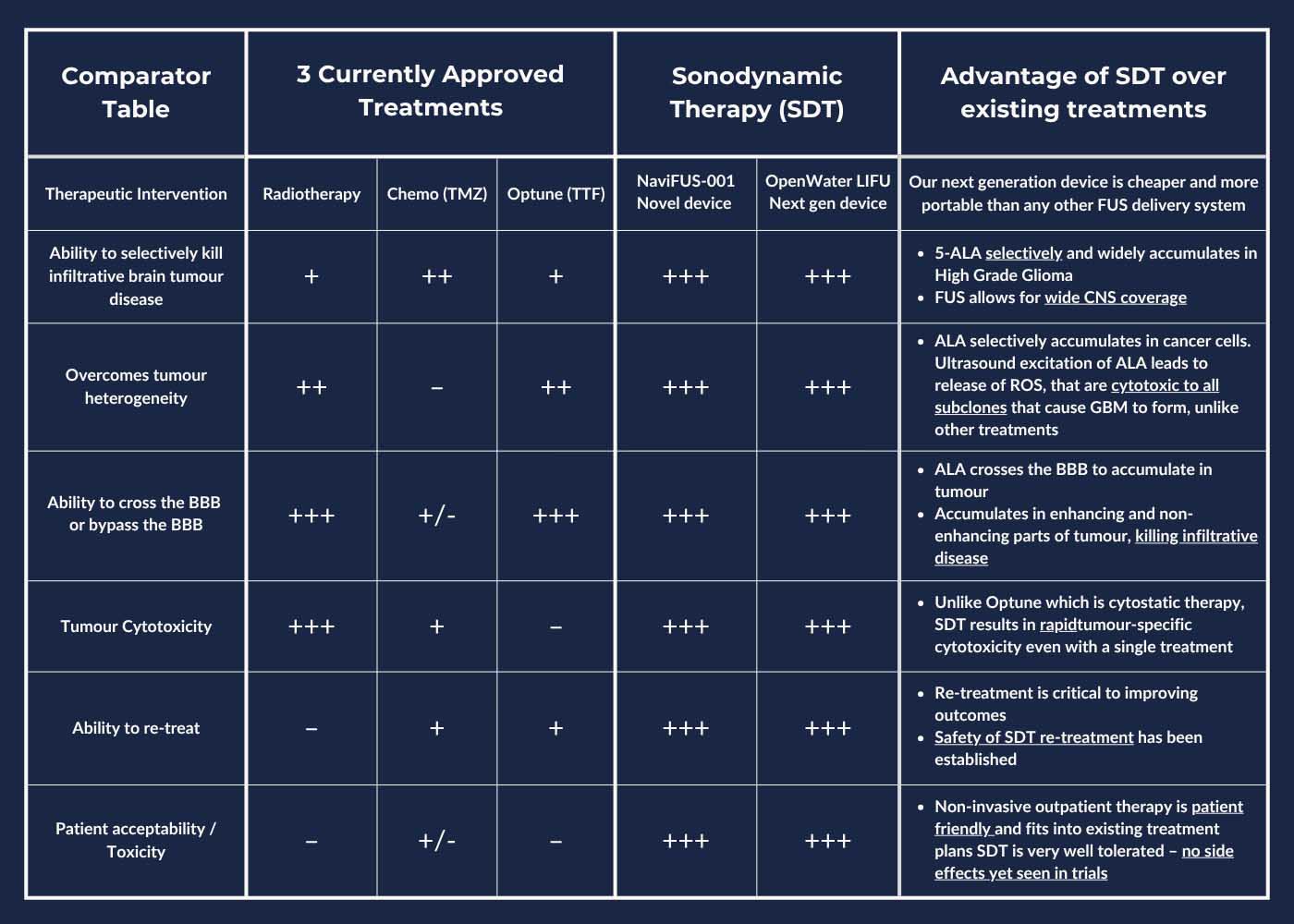
See annotated reference list below and tables further below comparing SDT to other approved treatments for GBM and comparing the new potential treatment device to the NaviUS, CV- 01 and Exablate FUS delivery devices.
SDT Webinar hosted by the Focused Ultrasound Foundation, September 28 2023:
https://www.fusfoundation.org/posts/focused-ultrasound-and-sonodynamic-therapy-webinar/
01
State of Practice on Transcranial MR-Guided Focused Ultrasound: A Report from the ASNR Standards and Guidelines Committee and ACR Commission on Neuroradiology Workgroup.
Shah, Tanabe, Jordan et al., State of Practice on Transcranial MR-Guided Focused Ultrasound: A Report from the ASNR Standards and Guidelines Committee and ACR Commission on Neuroradiology Workgroup. American Journal of Neuroradiology January 2025 46 (1) 2-10
02
A review / explanation of how 5-ALA is activated by Focused Ultrasound that results in the selective cell kill seen in Sonodynamic Therapy (SDT). It includes results from completed and ongoing preclinical and clinical studies, and outlines future directions for the technology.
Ebeling, A. & Prada, F. Sonodynamic Therapy Using 5-Aminolevulinic Acid for Malignant Gliomas: A Review. Life 2025, 15, 718.
03
Research paper demonstrating that the Cordance Cap prototype FUS delivery device can be used safely in patients with GBM (note the cap was used to successfully open the blood brain barrier in this study of 5 GBM patients):
Yuan J., Xu L., Chien CY. et al. First-in-human prospective trial of sonobiopsy in high-grade glioma patients using neuronavigation-guided focused ultrasound. npj Precis. Onc. 7, 92 (2023).
04
First clinical trial of SDT in recGBM. 12 patients recruited in an ‘energy dose escalation’ trial with three cohorts (200J, 400J and 800J). Half of the patients’ tumour was sonicated 5-7 days prior to complete tumour removal and histological analysis to look for pathological evidence of cell kill. The trial showed that SDT was safe, with no Adverse Events (AEs) due to the SDT therapy, andhistological analysis revealed that cell kill was achieved in the sonicated half of the tumour via reactive oxygen species release (i.e. the same mechanism of cell kill as photodynamic therapy).
Sanai N, Tovmasyan A, Tien AC, et al. An early clinical trial of 5-ALA sonodynamic therapy in recurrent high-grade glioma. Science Translational Medecine. 26 Nov 2025. Vol 17, Issue 826
05
First clinical trial results from use of an un-focused delivery device (CV-01 machine by Alpheus medical) that was used to deliver 3-4 SDT treatments one month apart to patients with recurrent high grade gliomas (NB: 2 of the 12 patients treated had recurrent Grade 3 oligodendroglioma, and 10 patients had recGBM): https://clinicaltrials.gov/study/NCT05362409
Although a safety trial, median survival of the treated patients was 15 months (as opposed to 6-8 months in historical controls). This is a greater improvement in overall survival than seen with tumour vaccines (e.g. DC-Vax), Tumour Treating Fields (Optune) or Bevacizumab (Avastin) – but note that these latter three experimental treatments have their survival benefit of 3-4 months calculated from adequately powered Phase 3 large clinical trials. Note that the CV-01 machine uses unfocused ultrasound and will therefore deliver less energy to deeper tumours than the Cordance Cap, which can deliver more energy deeply in brain (current theories suggest that higher energy delivery promotes greater cell kill).
Schulder M, Johns T, Mechtler L et al. Results from a phase I study of sonodynamic therapy with holohemispheric low-intensity non-ablative ultrasound in patients with recurrent high grade glioma. J.Neuro-onc (suppl 8): viii99.
https://academic.oup.com/neuro-oncology/article/26/Supplement_8/viii99/7889583
06
Safety publication from the first-in-human clinical trial of SDT for children with Diffuse Intrinsic Pontine Glioma (DIPG), also known as Diffuse Midline Glioma (DMG). This demonstrated no AEs or SAEs, with stabilisation of tumour growth. Subsequent unpublished presentations of this trial demonstrated reversal of neurological deficits and increase in median survival after Radiotherapy from 9 months to 16 months. This is unprecedented for DIPG / DMG, especially considering the trial was halted early and not all patients had all twelve of their planned monthly SDT treatments.
Syed HR, Kilburn L, Fonseca A, et al. First-in-human sonodynamic therapy with ALA for pediatric diffuse intrinsic pontine glioma: a phase 1/2 study using low-intensity focused ultrasound: technical communication. J Neurooncol. 2023; 162(2):449-451. https://clinicaltrials.gov/study/NCT05123534
07
Review of Sonodynamic Therapy for Neurosurgeons with some preliminary data from SonALAsense’s Phase 1/2 study of SDT for GBM in the Mayo, USA before the trial was terminated due to a lack of funds. No AEs or SAEs were noted in this trial, which was not powered to look at survival, nor had the repeat treatments from (4 and 5) above which demonstrated a doubling of survival time for recGBM and DIPG respectively. NB: The trial has re-opened for recruitment with repeat SDT treatments in August 2025 (see ref.10 vi below)
Hutton D, Burns T. and Hossain-Ibrahim K. Sonodynamic therapy for recurrent glioblastoma. Preliminary data from a Phase 2 energy dose escalation trial. Neurosurgery Focus (Sept 2024) 57(3), E7.
https://thejns.org/focus/view/journals/neurosurg-focus/57/3/article-pE7.xml
08
The first trial in Europe of SDT for recGBM to publish preliminary data, again showing no AEs or SAEs. (Note that the lead author has previously published data on the efficacy of PDT as a mechanism to treat GBM in a clinical trial setting – see Section 2.2 of reference 1 above – which we believe to be the same mechanism by which SDT works). This trial used monthly unfocused ultrasound treatments using the CV-01 machine:
Stummer W, Gerwing M, Bilgin SS, et al. Sonodynamic therapy with a single neoadjuvant, diffuse delivery of low-intensity ultrasound with 5-ALA in treatment naïve glioblastoma results in tumor-specific cytotoxic edema and increased apoptosis. J Neurooncol. 2025 May;172(3):687-693.
https://link.springer.com/article/10.1007/s11060-025-04957-7
09
The last trial to show a significant survival benefit for patients with GBM was in 2005 and the survival data within is often used as a comparator for ‘historic controls’. As a result of this trial radiotherapy and adjuvant temozolomide chemotherapy is now standard of care for GBM, after maximal safe surgical debulking / resection (the Stupp Protocol):
Stupp R. et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med (2005) 352, 987-996, doi:10.1056/NEJMoa043330
10
Review paper discussing the possibility that SDT can have an abscopal effect – i.e. the antigen release from the cell kill causes an ‘auto-vaccination’ effect, as has been seen in PDT, and greater survival benefit than can be explained by the sonication field itself:
Collins VG, Hutton D, Hossain-Ibrahim K and Banerjee S. The abscopal effects of sonodynamic therapy in cancer. Br J Cancer (2025) 132: 409-420
11
Other clinical trials of SDT for glioblastoma that have either not yet been published or are still recruiting:
i. SonicALA clinical trial of SDT for newly diagnosed GBM prior to resection (safety study, Milan, Italy). This trial used the more invasive Insightec Exablate machine that requires skull pins to place a fixed helmet on the patient, with treatment performed lying still inside an MRI scanner (i.e. requiring considerably more resources, time and cost compared to our new potential treatment device – see comparator table below); https://www.clinicaltrials.gov/study/NCT04845919
ii. Continuation of Nader Sanai’s safety study (ref 2 above) to expand to 45 patients to gain preliminary efficacy data in patients with recGBM:
https://clinicaltrials.gov/study/NCT04559685
iii. The first safety and feasibility study to investigate the use of the NaviFUS machine to deliver SDT to patients with recGBM (see comparator table below): https://clinicaltrials.gov/study/NCT06039709
iv. Trial of unfocused ultrasound with CV-01 device 2-5 days prior to surgical resection of GBM to evaluate safety and early histological effects (preliminary data published in reference 7 above): https://clinicaltrials.gov/study/NCT06665724
v. Phase 2 study of Alpheus Medical’s Low Intensity Diffuse (unfocused) Ultrasound for recurrent high grade gliomas (using the CV-01 machine – see comparator table below): Not yet registered with clinicaltrials.gov
vi. Phase 1b trial to test safety, best dose and preliminary efficacy data (n=8) of intravenous ALA with the Insightec Exablate MR-guided FUS device. Treatments are to be repeated every 6 weeks until disease progression or unacceptable toxicity: https://clinicaltrials.gov/study/NCT07076472
vii. First Randomised 1:1 trial to compare SDT + Standard of Care to SOC alone (radiotherapy, temozolomide and bevacizumab) for patients with newly diagnosed GBM, in Zenghzhou, China (2025). Aiming to recruit 220 patients, using intra-arterial Hiporfin® as the sonosensitiser (which requires avoidance of sunlight for 30 days) and treatments every day for 3 days followed by 24 mths monitoring. Note Phase 1 study not published: https://clinicaltrials.gov/study/NCT07130149
viii. First Randomised 1:1 trial to compare SDT + Standard of Care to SOC alone (radiotherapy, temozolomide and bevacizumab) for patients with DIPG, in Zenghzhou, China (2025). Aiming to recruit 216 patients using intravenous Hematoporphyrin (‘Xipofen’) twice per day for 4 days. Note that their Phase 1 study showed only a two month survival benefit and there is little data published on their handheld ultrasound device: https://clinicaltrials.gov/study/NCT06999148
Comparison of potential new treatment device with SDT to Chemotherapy, Radiotherapy, and Optune (Tumour Treating Fields), which demonstrated 3-4 month extra survival in GBM.